These are the relative volumes of the molecules. Site blocking gives rise to large nonideal entropy of mixing and it can only be estimated in multicomponent systems through the use of Monte Carlo simulations, 9 empirical methods or by computing the partial entropy of mixing as proposed by Thorn and Wisnlow 10,11 The more complex CVM is limited by practical reasons to a low number of. 
The result is an equation for the Gibbs free energy change Δ G m i x, there is separation into two coexisting phases, one richer in polymer but poorer in solvent, than the other. Three factors which may contribute to the excess entropy of mixing of binary solutions have been explored. The ideal solution was introduced as a mixture with zero enthalpy of mixing H 0 and ideal entropy of mixing IS S RT Xi ln Xi. This deviation gives rise to excess heat capacities of mixing and in consequence to excess vibrational entropies of mixing. The entropy contribution due to lattice vibrations. On collecting the different contributions to the excess entropy of mixing, we obtain S xs S xs,conf + S vib + S el + S mag Sex,conf has been discussed in the previous section and the relevant expression is given in Eq. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart. Simple scheme for the excess entropy of mixing. Introduction to Chemical Engineering Thermodynamics. There is more mixing involved, but the atoms of the gas go. 
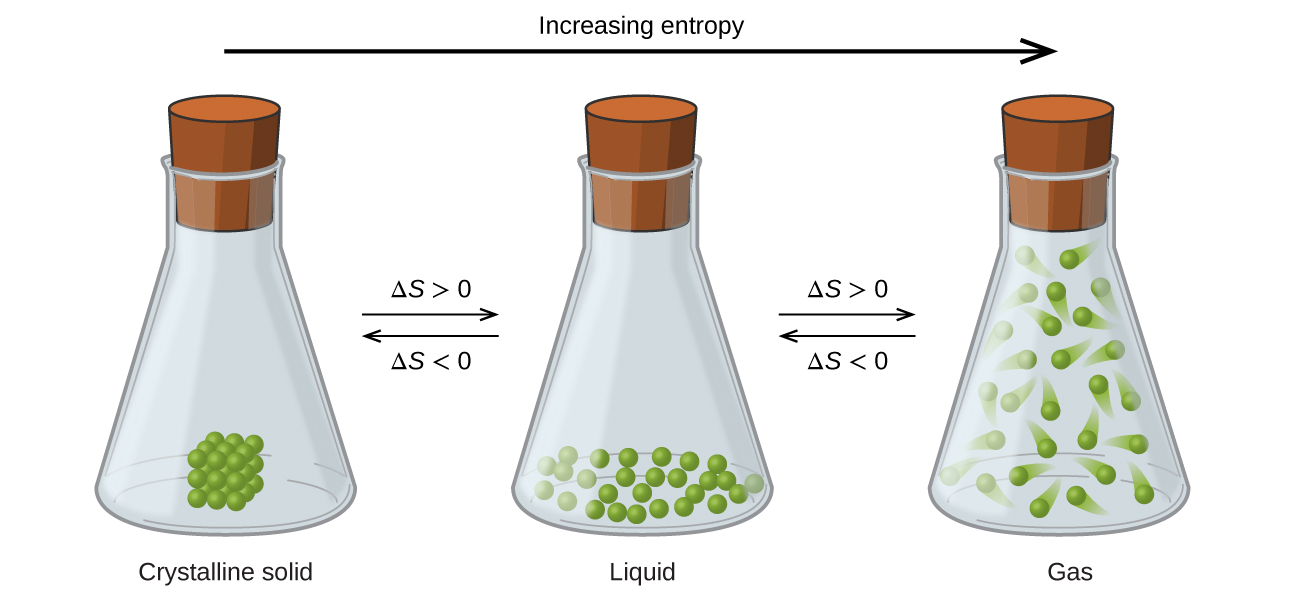
nA over (nA+nB) times R log (VA over (VA+. Examining the mixing process on a molecular. Entropy usually decreases when a gas dissolves in a liquid or solid. The molar entropy of mixing is total entropy change divided by the total moles. (5.28) and the molar Gibbs ideal mixing energy is. The number of permutations which are possible, assuming ideal interchanges within each sublattice, is given by the following equation. \) between adjacent sites of the lattice.Lattice model of polymer solutions Mixture of polymers and solvent on a lattice.įlory–Huggins solution theory is a lattice model of the thermodynamics of polymer solutions which takes account of the great dissimilarity in molecular sizes in adapting the usual expression for the entropy of mixing. In solid solutions, the configurational entropy is unequal to zero and the heat capacity versus composition behaviour generally deviates from a linear relationship, especially at low temperatures of 50 K. What is the entropy of mixing of 1 mol of O2 with 1 mole of N2 at 25oC, assuming that they are ideal gases final answer: 11.53 J K-1. Equation (7.1) states that there is an entropy increase due to the increased volume that each gas is able to access. The ideal entropy of mixing is made up of the configurational contributions by components mixing on each of the sublattices.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
