
In the α anomer, the -OH substituent on the anomeric carbon rests on the opposite side (trans) of the ring from the CH2OH side branch. The resulting possible pair of stereoisomers are called anomers. Rings with five and six atoms are called furanose and pyranose forms, respectively, and exist in equilibrium with the straight-chain form.ĭuring the conversion from straight-chain form to the cyclic form, the carbon atom containing the carbonyl oxygen, called the anomeric carbon, becomes a stereogenic center with two possible configurations: The oxygen atom may take a position either above or below the plane of the ring. The aldehyde or ketone group of a straight-chain monosaccharide will react reversibly with a hydroxyl group on a different carbon atom to form a hemiacetal or hemiketal, forming a heterocyclic ring with an oxygen bridge between two carbon atoms. This usage of "d-" and "l-" is no longer followed in carbohydrate chemistry. The "D-" and "L-" prefixes should not be confused with "d-" or "l-", which indicate the direction that the sugar rotates plane polarized light. 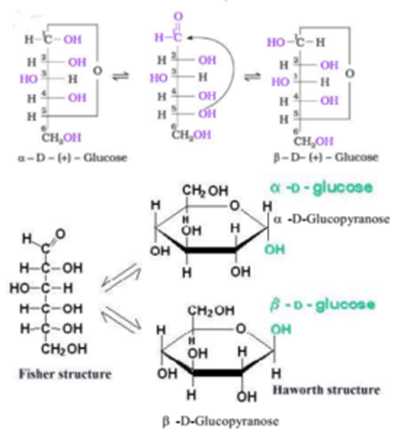
The assignment of D or L is made according to the orientation of the asymmetric carbon furthest from the carbonyl group: in a standard Fischer projection if the hydroxyl group is on the right the molecule is a D sugar, otherwise it is an L sugar. In the case of glyceraldehyde, an aldotriose, there is one pair of possible stereoisomers, which are enantiomers and epimers.ġ,3-dihydroxyacetone, the ketose corresponding to the aldose glyceraldehyde, is a symmetric molecule with no stereocenters). H2O)6, of which all but two of its six carbons atoms are stereogenic, making D-glucose one of 2x2x2x2 = 16 possible stereoisomers.The aldohexose D-glucose, for example, has the formula (C For example, glucose is an aldohexose (a six-carbon aldehyde), ribose is an aldopentose (a five-carbon aldehyde), and fructose is a ketohexose (a six-carbon ketone).Įach carbon atom bearing a hydroxyl group (-OH), with the exception of the first and last carbons, are asymmetric, making them stereocenters with two possible configurations each (R or S).īecause of this asymmetry, a number of isomers may exist for any given monosaccharide formula. These two systems of classification are often combined. Monosaccharides with three carbon atoms are called trioses, those with four are called tetroses, five are called pentoses, six are hexoses, and so on. If the carbonyl group is an aldehyde, the monosaccharide is an aldose if the carbonyl group is a ketone, the monosaccharide is a ketose. Monosaccharides are classified according to three different characteristics: deoxy sugar (fucose, Fuculose, Rhamnose).aldohexose (Allose, Altrose, glucose, mannose, Gulose, Idose, galactose, Talose).ketohexose (Psicose, fructose, Sorbose, Tagatose).Aldopentose (Ribose, Arabinose, Xylose, Lyxose).Monosaccharides are the building blocks of disaccharides like sucrose (common sugar) and polysaccharides (such as cellulose and starch).įurther, each carbon atom that supports a hydroxyl group (except for the first and last) is chiral, giving rise to a number of isomeric forms all with the same chemical formula.įor instance, galactose and glucose are both aldohexoses, but they have different chemical and physical properties. In plants, starch is used for the same purpose.Įxamples of monosaccharides include glucose (dextrose), fructose, galactose, xylose and ribose. In many animals, including humans, this storage form is glycogen, especially in liver and muscle cells. When monosaccharides are not immediately needed by many cells they are often converted to more space efficient forms, often polysaccharides. Monosaccharides are the major source of fuel for metabolism, being used both as an energy source ( glucose being the most important in nature) and in biosynthesis. They consist of one sugar and are usually colorless, water-soluble, crystalline solids. They cannot be hydrolyzed into simpler sugars. Definition: Monosaccharides (from Greek monos: single, sacchar: sugar) are the simplest carbohydrates.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
